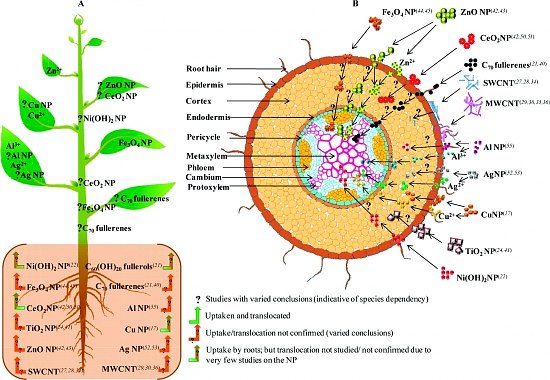
I’ve dug up more information on nanomaterials and regulatory frameworks but before I launch off into the discussion I think it might be interesting to take a look at this graphic of a plant’s potential uptake of various nanomaterials as it illustrates some of the reasons why there’s so much interest in this topic.
Downloaded from the June 7, 2011 article, Nano & The Food Chain: Another Puzzle by Gwyneth K. Shaw for the New Haven Independent (the graphic was originally published in the Journal of Agricultural and Food Chemistry),
Shaw’s article is about a study (Interaction of Nanoparticles with Edible Plants and Their Possible Implications in the Food Chain [this is behind a paywall]) by researchers at the University of Texas at El Paso, which reviews current studies in the field and suggests that as nanoparticles enter the food chain we need to consider cumulative effects.
Meanwhile, the discussion about developing regulatory frameworks and whether or not we need to have a definition for nanomaterials before setting a regulatory framework continues. From the June 7, 2011 news item on Nanowerk,
The Belgian Presidency of the Council of the European Union organized a high level event on September 14, 2010, bringing together representatives of various associations (consumers, environmental protection, workers, industrial federations), scientists, regulatory experts as well as national and European regulatory bodies, in order to review the legislative initiatives in progress with regard to nanomaterials and to establish an operational framework for the management of incidents in the short term and to achieve improved risk management in the long term.
Initially I confused this meeting with the March 2011 meeting mentioned in my April 14, 2011 posting but I gather there are a number of meetings (some of which seem remarkably similar) on the topic with various European Union groups and subgroups. The September 2010 meeting was under the auspices of the European Union and the March 2011 meeting was under the auspices of the European Commission (which I believe is part of the European Union bureaucracy). In any event, the September 2010 meeting resulted in a set of objectives being set (from the news item),
THE [European Union] PRESIDENCY CONCLUDES THAT, IN ORDER TO protect the workers, consumers health and the environment, and at the same time guarantee the development of a secure and sound economy based notably on innovation and societally acceptable industrial applications that create quality jobs, THE FOLLOWING OBJECTIVES MUST BE REACHED, IN RELATION TO NANOMATERIALS, PRODUCTS CONTAINING NANOMATERIALS AND NANOTECHNOLOGIES:
1) REGARDING THE REGULATORY FRAMEWORK:
- to effectively address their potential risks and uncertainties, at the earliest, and thus ensure a high level of environment and health protection;
- to consider their challenges transversally, across sectors, disciplines and regulations;
- in parallel, to implement specific regulatory measures to deal with their particularities;
- to appropriately inform and consult consumers, workers and citizens;
2) REGARDING SCIENCE, RESEARCH, INNOVATION AND KNOWLEDGE:
- to develop the necessary scientific knowledge in a global, coordinated and open manner;
- to be proactive and to anticipate when dealing with the risks and uncertainties of new technological developments.
IN CONSEQUENCE, THE FOLLOWING ACTIONS HAVE TO BE TAKEN:
- to take up responsibilities at the Member States level and, during a transitory period, draw up coordinated and integrated national strategies and concrete measures in favour of risk management, information and monitoring;
- to develop urgently a regulatory definition for nanomaterials that must include nanomaterials all along their lifecycle, including into substances, products, articles, wearing residues and waste; [emphasis mine]
- to consider nanotechnology as a priority into a future 2nd Environment and Health Action Plan, including inter alia basic and applied research related to them, their specific potential risks, their traceability and the link between innovation, environment and health safety;
- to clarify the various issues that remain presently unaddressed in the Commission proposals to adapt REACH to the nanomaterials and, in addition to the adaptations to the guidances to include significant modifications into the REACH 2012 review, including the lowering of the tonnage triggers for nanomaterials, modifications to data requirements in REACH annexes, consideration of nanomaterials as new substances, annexes V (exemptions) and XIII review (PBT, vPvB) and the inclusion in REACH of a definition of nanomaterials and articles containing nanomaterials;
- to increase public and private resources, especially the financial inputs to the OECD WPMN, with the goal of obtaining results to be used for regulatory purposes as soon as possible;
- to develop harmonized compulsory databases of nanomaterials and products containing nanomaterials;
- such databases must be the base for traceability, market surveillance, gaining knowledge for better risk prevention and for the improvement of the legislative framework;
- to take into account, in the design of such databases, the need for providing information to the citizens, workers and consumers regarding nanomaterials and products containing nanomaterials as well as the industry’s need for data protection;
- claims made on labels of products containing nanomaterials must be regulated and the requirements to inform the consumer of the presence of nanomaterials in consumer products must be defined;
- to consider sustainability, societal benefits, demands for public participation, and ethical considerations in the public investments in innovative technologies;
- to establish a systematic, balanced and appropriate link between on the one hand the assessment of risk, early warnings and uncertainties and on the other hand the public investments in innovative technologies in general and nanotechnologies in particular, including financing mechanisms that take such a link into account;
- to consider research in toxicology and ecotoxicology of nanomaterials, as well as their fate along the whole lifecycle as a high priority.
There is a school of thought that a regulatory framework can be put in place without establishing a definition beforehand as per my April 15, 2011 posting where I mentioned Dr. Andrew Maynard’s proposal and expressed some hesitation. I see Dexter Johnson (of the Nanoclast blog on the IEEE [Institute of Electrical and Electronics Engineers] website), after interviewing Rudolf Strohmeier, Deputy Director General, Directorate General for Research & Innovation for the European Commission at the EuroNano Forum 2011 in Budapest, Hungary, has weighed in with this in his May 31, 2011 posting,
Below is an audio recording I made of my exchange with Mr. Strohmeier. Interestingly, according to him, the definition was necessary for educating EU citizens as much as for developing regulations. …
In fairness, I didn’t really get a chance to follow up with Mr. Strohmeier to see if he could see the problems that arise when you arbitrarily arrive at a definition that may not always reflect the latest science on the topic. Nonetheless, I can’t help but think that a definition that is as much about mollifying the public as it is about good science has inherent risks itself. [emphases mine]
I take Dexter’s and Andrew’s point about the potential problems that creating a definition for what I’m going to call ‘public relations purposes’ could cause but I still haven’t grasped how one would create a regulatory framework without a definition of some kind (but maybe that’s just the writer in me).
All of this certainly puts the Canadian situation into perspective. There’s an interim definition in place. As for a regulatory framework, it appears that the government (Health Canada) favours a case by case approach as per their plans to investigate nanosunscreens (noted in my June 3, 2011 posting).
Pingback: The EC Defines a Nanomaterial: Now What? | My Blog