If this works out, it would make testing for COVID-19 an infinitely easier task. From a May 29, 2020 news item on phys.org,
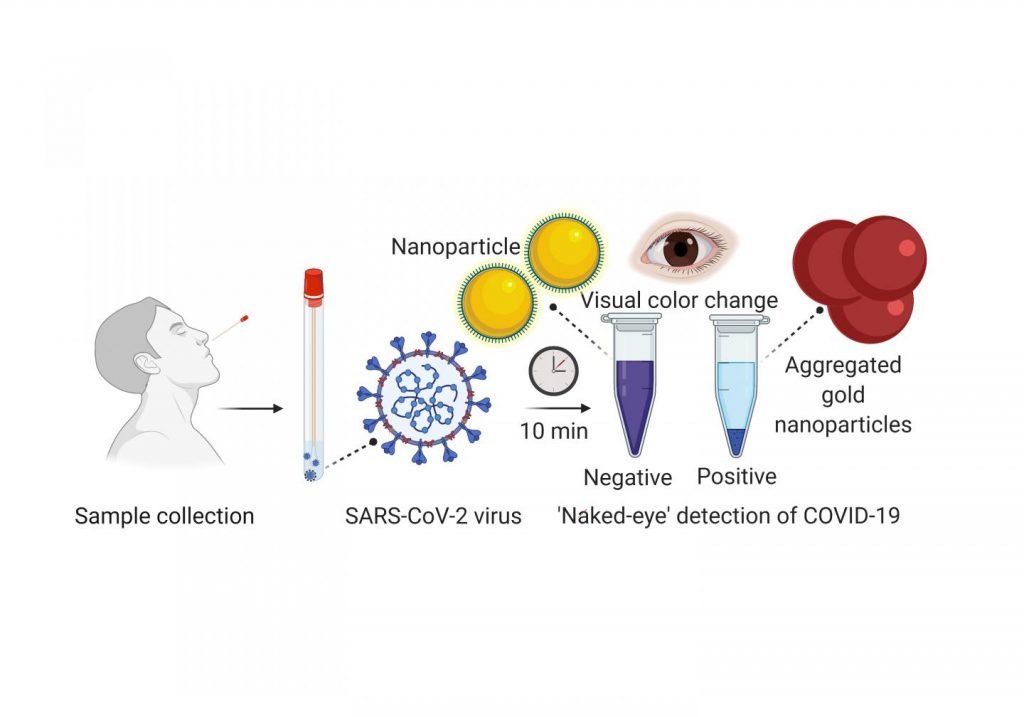
Scientists from the University of Maryland School of Medicine (UMSOM) developed an experimental diagnostic test for COVID-19 that can visually detect the presence of the virus in 10 minutes. It uses a simple assay containing plasmonic gold nanoparticles to detect a color change when the virus is present. The test does not require the use of any advanced laboratory techniques, such as those commonly used to amplify DNA, for analysis. The authors published their work last week [May 21, 2020] in the American Chemical Society’s nanotechnology journal ACS Nano.
“Based on our preliminary results, we believe this promising new test may detect RNA [ribonucleic acid] material from the virus as early as the first day of infection. Additional studies are needed, however, to confirm whether this is indeed the case,” said study leader Dipanjan Pan, PhD, Professor of Diagnostic Radiology and Nuclear Medicine and Pediatrics at the UMSOM.
A May 28, 2020 University of Maryland news release (also on EurekAlert), which originated the news item, provides more detail,
Once a nasal swab or saliva sample is obtained from a patient, the RNA is extracted from the sample via a simple process that takes about 10 minutes. The test uses a highly specific molecule attached to the gold nanoparticles to detect a particular protein. This protein is part of the genetic sequence that is unique to the novel coronavirus. When the biosensor binds to the virus’s gene sequence, the gold nanoparticles respond by turning the liquid reagent from purple to blue.
“The accuracy of any COVID-19 test is based on being able to reliably detect any virus. This means it does not give a false negative result if the virus actually is present, nor a false positive result if the virus is not present,” said Dr. Pan. “Many of the diagnostic tests currently on the market cannot detect the virus until several days after infection. For this reason, they have a significant rate of false negative results.”
Dr. Pan created a company called VitruVian Bio to develop the test for commercial application. He plans to have a pre-submission meeting with the U.S. Food and Drug Administration (FDA) within the next month to discuss requirements for getting an emergency use authorization for the test. New FDA policy allows for the marketing of COVID-19 tests without requiring them to go through the usual approval or clearance process. These tests do, however, need to meet certain validation testing requirements to ensure that they provide reliable results.
“This RNA-based test appears to be very promising in terms of detecting the virus. The innovative approach provides results without the need for a sophisticated laboratory facility,” said study co-author Matthew Frieman, PhD, Associate Professor of Microbiology and Immunology at UMSOM.
Although more clinical studies are warranted, this test could be far less expensive to produce and process than a standard COVID-19 lab test; it does not require laboratory equipment or trained personnel to run the test and analyze the results. If this new test meets FDA expectations, it could potentially be used in daycare centers, nursing homes, college campuses, and work places as a surveillance technique to monitor any resurgence of infections.
In Dr. Pan’s laboratory, research scientist Parikshit Moitra, PhD, and UMSOM research fellow Maha Alafeef conducted the studies along with research fellow Ketan Dighe from UMBC.
Dr. Pan holds a joint appointment with the College of Engineering at the University of Maryland Baltimore County and is also a faculty member of the Center for Blood Oxygen Transport and Hemostasis (CBOTH).
“This is another example of how our faculty is driving innovation to fulfill a vital need to expand the capacity of COVID-19 testing,” said Dean E. Albert Reece, MD, PhD, MBA, who is also Executive Vice President for Medical Affairs, UM Baltimore, and the John Z. and Akiko K. Bowers Distinguished Professor, University of Maryland School of Medicine. “Our nation will be relying on inexpensive, rapid tests that can be dispersed widely and used often until we have effective vaccines against this pandemic.”
Here’s a link to and a citation for the paper,
Selective Naked-Eye Detection of SARS-CoV-2 Mediated by N Gene Targeted Antisense Oligonucleotide Capped Plasmonic Nanoparticles by Parikshit Moitra, Maha Alafeef, Ketan Dighe, Matthew B. Frieman, and Dipanjan Pan. ACS Nano 2020, XXXX, XXX, XXX-XXX DOI: https://doi.org/10.1021/acsnano.0c03822 Publication Date:May 21, 2020 Copyright © 2020 American Chemical Society
This paper appears to be open access.
I tried to find Dr. Pan’s company, VitruVian Bio and found a business with an almost identical name, Vitruvian Biomedical, which does not include Dr. Pan on its management team list and this company’s focus is on Alzheimer’s Disease. Finally, there is no mention of the COVID-19 test anywhere on the Vitruvian Biomedical website.