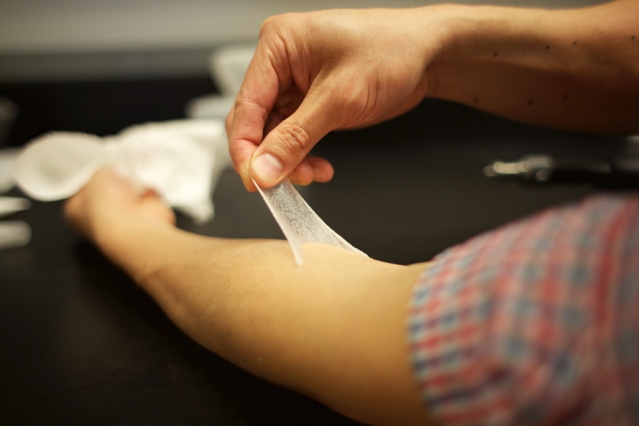
“It’s an invisible layer that can provide a barrier, provide cosmetic improvement, and potentially deliver a drug locally to the area that’s being treated. Those three things together could really make it ideal for use in humans,” Daniel Anderson says. Photo: Melanie Gonick/MIT
It almost looks like he’s peeling off his own skin and I imagine that’s the secret to this polymer’s success. A May 9, 2016 news item on phys.org describes the work being done at the Massachusetts Institute of Technology (MIT) and elsewhere with collaborators,
Scientists at MIT, Massachusetts General Hospital, Living Proof, and Olivo Labs have developed a new material that can temporarily protect and tighten skin, and smooth wrinkles. With further development, it could also be used to deliver drugs to help treat skin conditions such as eczema and other types of dermatitis.
A May 9, 2016 MIT news release (also on EurekAlert), which originated the news item, provides more detail,
The material, a silicone-based polymer that could be applied on the skin as a thin, imperceptible coating, mimics the mechanical and elastic properties of healthy, youthful skin. In tests with human subjects, the researchers found that the material was able to reshape “eye bags” under the lower eyelids and also enhance skin hydration. This type of “second skin” could also be adapted to provide long-lasting ultraviolet protection, the researchers say.
“It’s an invisible layer that can provide a barrier, provide cosmetic improvement, and potentially deliver a drug locally to the area that’s being treated. Those three things together could really make it ideal for use in humans,” says Daniel Anderson, an associate professor in MIT’s Department of Chemical Engineering and a member of MIT’s Koch Institute for Integrative Cancer Research and Institute for Medical Engineering and Science (IMES).
Anderson is one of the authors of a paper describing the polymer in the May 9 online issue of Nature Materials. Robert Langer, the David H. Koch Institute Professor at MIT and a member of the Koch Institute, is the paper’s senior author, and the paper’s lead author is Betty Yu SM ’98, ScD ’02, former vice president at Living Proof. Langer and Anderson are co-founders of Living Proof and Olivo Labs, and Yu earned her master’s and doctorate at MIT.
Mimicking skin
As skin ages, it becomes less firm and less elastic — problems that can be exacerbated by sun exposure. This impairs skin’s ability to protect against extreme temperatures, toxins, microorganisms, radiation, and injury. About 10 years ago, the research team set out to develop a protective coating that could restore the properties of healthy skin, for both medical and cosmetic applications.
“We started thinking about how we might be able to control the properties of skin by coating it with polymers that would impart beneficial effects,” Anderson says. “We also wanted it to be invisible and comfortable.”
The researchers created a library of more than 100 possible polymers, all of which contained a chemical structure known as siloxane — a chain of alternating atoms of silicon and oxygen. These polymers can be assembled into a network arrangement known as a cross-linked polymer layer (XPL). The researchers then tested the materials in search of one that would best mimic the appearance, strength, and elasticity of healthy skin.
“It has to have the right optical properties, otherwise it won’t look good, and it has to have the right mechanical properties, otherwise it won’t have the right strength and it won’t perform correctly,” Langer says.
The best-performing material has elastic properties very similar to those of skin. In laboratory tests, it easily returned to its original state after being stretched more than 250 percent (natural skin can be elongated about 180 percent). In laboratory tests, the novel XPL’s elasticity was much better than that of two other types of wound dressings now used on skin — silicone gel sheets and polyurethane films.
“Creating a material that behaves like skin is very difficult,” says Barbara Gilchrest, a dermatologist at MGH and an author of the paper. “Many people have tried to do this, and the materials that have been available up until this have not had the properties of being flexible, comfortable, nonirritating, and able to conform to the movement of the skin and return to its original shape.”
The XPL is currently delivered in a two-step process. First, polysiloxane components are applied to the skin, followed by a platinum catalyst that induces the polymer to form a strong cross-linked film that remains on the skin for up to 24 hours. This catalyst has to be added after the polymer is applied because after this step the material becomes too stiff to spread. Both layers are applied as creams or ointments, and once spread onto the skin, XPL becomes essentially invisible.
High performance
The researchers performed several studies in humans to test the material’s safety and effectiveness. In one study, the XPL was applied to the under-eye area where “eye bags” often form as skin ages. These eye bags are caused by protrusion of the fat pad underlying the skin of the lower lid. When the material was applied, it applied a steady compressive force that tightened the skin, an effect that lasted for about 24 hours.
In another study, the XPL was applied to forearm skin to test its elasticity. When the XPL-treated skin was distended with a suction cup, it returned to its original position faster than untreated skin.
The researchers also tested the material’s ability to prevent water loss from dry skin. Two hours after application, skin treated with the novel XPL suffered much less water loss than skin treated with a high-end commercial moisturizer. Skin coated with petrolatum was as effective as XPL in tests done two hours after treatment, but after 24 hours, skin treated with XPL had retained much more water. None of the study participants reported any irritation from wearing XPL.
“I think it has great potential for both cosmetic and noncosmetic applications, especially if you could incorporate antimicrobial agents or medications,” says Thahn Nga Tran, a dermatologist and instructor at Harvard Medical School, who was not involved in the research.
Living Proof has spun out the XPL technology to Olivo Laboratories, LLC, a new startup formed to focus on the further development of the XPL technology. Initially, Olivo’s team will focus on medical applications of the technology for treating skin conditions such as dermatitis.
This video supplied by MIT shows how to apply the polymer and offers a description and demonstration of its properties once applied,
Here’s a link to and a citation for the paper,
An elastic second skin by Betty Yu, Soo-Young Kang, Ariya Akthakul, Nithin Ramadurai, Morgan Pilkenton, Alpesh Patel, Amir Nashat, Daniel G. Anderson, Fernanda H. Sakamoto, Barbara A. Gilchrest, R. Rox Anderson & Robert Langer. Nature Materials (2016) doi:10.1038/nmat4635 Published online 09 May 2016
This paper is behind a paywall.
One final comment, I wonder who’s lining up to invest in this product.