Michael Berger in his March 16, 2015 Nanowerk Spotlight article profiles some very interesting research into replacing chemicals with water nanostructures,
The burden of foodborne diseases worldwide is huge, with serious economic and public health consequences. The CDC [US Centers for Disease Control] estimates that each year in the USA approximately 48 million people get sick, 128,000 get hospitalized and 3,000 die from the consumption of food contaminated with pathogenic microorganisms. The food industry is in search of effective intervention methods that can be applied from ‘farm to fork’ to ensure the safety of the food chain and be consumer and environment friendly at the same time.
In the food industry, chemicals are routinely used to clean and disinfect product contact surfaces as well as the outer surface of the food itself. These chemicals provide a necessary and required step to ensure that the foods produced and consumed are as free as possible from microorganisms that can cause foodborne illness.
Food activists are concerned that some of the chemicals used by the food industry for disinfection can cause health issues for consumers. A prime example is the current discussion in Europe about ‘American chlorine chicken’. …
Berger goes on to highlight the research being conducted at the Harvard T. Chan School of Public Health (Harvard University). The team announced a new technique called Engineered Water Nanostructures (EWNS), which is generated by electrospraying water. The team published this paper in 2014,
A chemical free, nanotechnology-based method for airborne bacterial inactivation using engineered water nanostructures by Georgios Pyrgiotakis, James McDevitt, Andre Bordini, Edgar Diaz, Ramon Molina, Christa Watson, Glen Deloid, Steve Lenard, Natalie Fix, Yosuke Mizuyama, Toshiyuki Yamauchi, Joseph Brain and Philip Demokritou. Environ. Sci.: Nano, 2014,1, 15-26 DOI: 10.1039/C3EN00007A
First published online 28 Nov 2013
This paper is open access.
More recently, the team has proved the efficacy of this technique on stainless steel surfaces and tomatoes. A Feb. 25, 2015 Harvard T. Chan School of Public Health news release provides information about the costs of foodborne diseases and goes on to describe the technique and the latest experiments,
The burden of foodborne diseases worldwide is huge, with serious economic and public health consequences. The U.S. Department of Agriculture’s (USDA’s) Economic Research Service reported in 2014 that foodborne illnesses are costing the economy more than $15.6 billion and about 53,245 Americans visit the hospital annually due to foodborne illnesses. The food industry is in search of effective intervention methods that can be applied form “farm to fork” to ensure the safety of the food chain and be consumer and environment friendly at the same time.
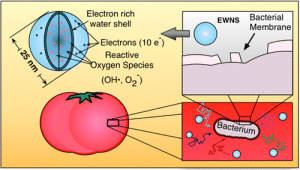
Researchers at the Center for Nanotechnology and Nanotoxicology of the Harvard T. Chan School of Public Health are currently exploring the effectiveness of a nanotechnology based, chemical free, intervention method for the inactivation of foodborne and spoilage microorganisms on fresh produce and on food production surfaces. This method utilizes Engineered Water Nanostructures (EWNS) generated by electrospraying of water. EWNS possess unique properties; they are 25 nm in diameter, remain airborne in indoor conditions for hours, contain Reactive Oxygen Species (ROS), have very strong surface charge (on average 10e/structure) and have the ability to interact and inactivate pathogens by destroying their membrane.
In a study funded by the USDA and just published this week in the premier Environmental Science and Technology journal, the efficacy of these tiny water nanodroplets, in inactivating representative foodborne pathogens such as Escherichia coli, Salmonella enterica and Listeria innocua, on stainless steel surfaces and on tomatoes, was assessed showing significant log reductions in inactivation of select food pathogens. These promising results could open up the gateway for further exploration into the dynamics of this method in the battle against foodborne disease. More importantly this novel, chemical-free, cost effective and environmentally friendly intervention method holds great potential for development and application in the food industry, as a ‘green’ alternative to existing inactivation methods.
Here’s a link to and a citation for the latest paper,
Inactivation of Foodborne Microorganisms Using Engineered Water Nanostructures (EWNS) by Georgios Pyrgiotakis, Archana Vasanthakumar, Ya Gao, Mary Eleftheriadou, Eduardo Toledo, Alice DeAraujo, James McDevitt, Taewon Han, Gediminas Mainelis, Ralph Mitchell, and Philip Demokritou. Environ. Sci. Technol., Article ASAP DOI: 10.1021/es505868a Publication Date (Web): February 19, 2015
Copyright © 2015 American Chemical Society
This paper is behind a paywall. The researchers have made this image illustrating a ‘water shell’s’ effect on a bacterium located on a tomato,
I’m not sure how chemical companies are going to feel but this is very exciting news. Still, one has to wonder just how much water this technique would require for full scale adoption and would it be reusable?