A fascinating new use for hyaluronic acid (usually discussed in relation to cosmetic wrinkle-reduction) has been found according to a November 1, 2023 news item on ScienceDaily.
In a recent publication in the journal Nature, researchers from the Institute of Basic Science (IBS) in South Korea have made significant strides in biomaterial technology and rehabilitation medicine. They’ve developed a novel approach to healing muscle injury by employing “injectable tissue prosthesis” in the form of conductive hydrogels and combining it with a robot-assisted rehabilitation system.
Let’s imagine you are swimming in the ocean. A giant shark approaches and bites a huge chunk of meat out of your thigh, resulting in a complete loss of motor/sensor function in your leg.
If left untreated, such severe muscle damage would result in permanent loss of function and disability.
How on Earth will you be able to recover from this kind of injury?
Traditional rehabilitation methods for these kinds of muscle injuries have long sought an efficient closed-loop gait rehabilitation system that merges lightweight exoskeletons and wearable/implantable devices.
Such assistive prosthetic system is required to aid the patients through the process of recovering sensory and motor functions linked to nerve and muscle damage.
Unfortunately, the mechanical properties and rigid nature of existing electronic materials render them incompatible with soft tissues.
This leads to friction and potential inflammation, stalling patient rehabilitation.
To overcome these limitations, the IBS researchers turned to a material commonly used as a wrinkle-smoothing filler, called hyaluronic acid.
…
A November 2, 2023 Institute of Basic Science (IBS) press release (also on EurekAlert but published November 1, 2023), which originated the news item, explains how hyaluronic acid helps in tissue rehabilitation and regeneration,
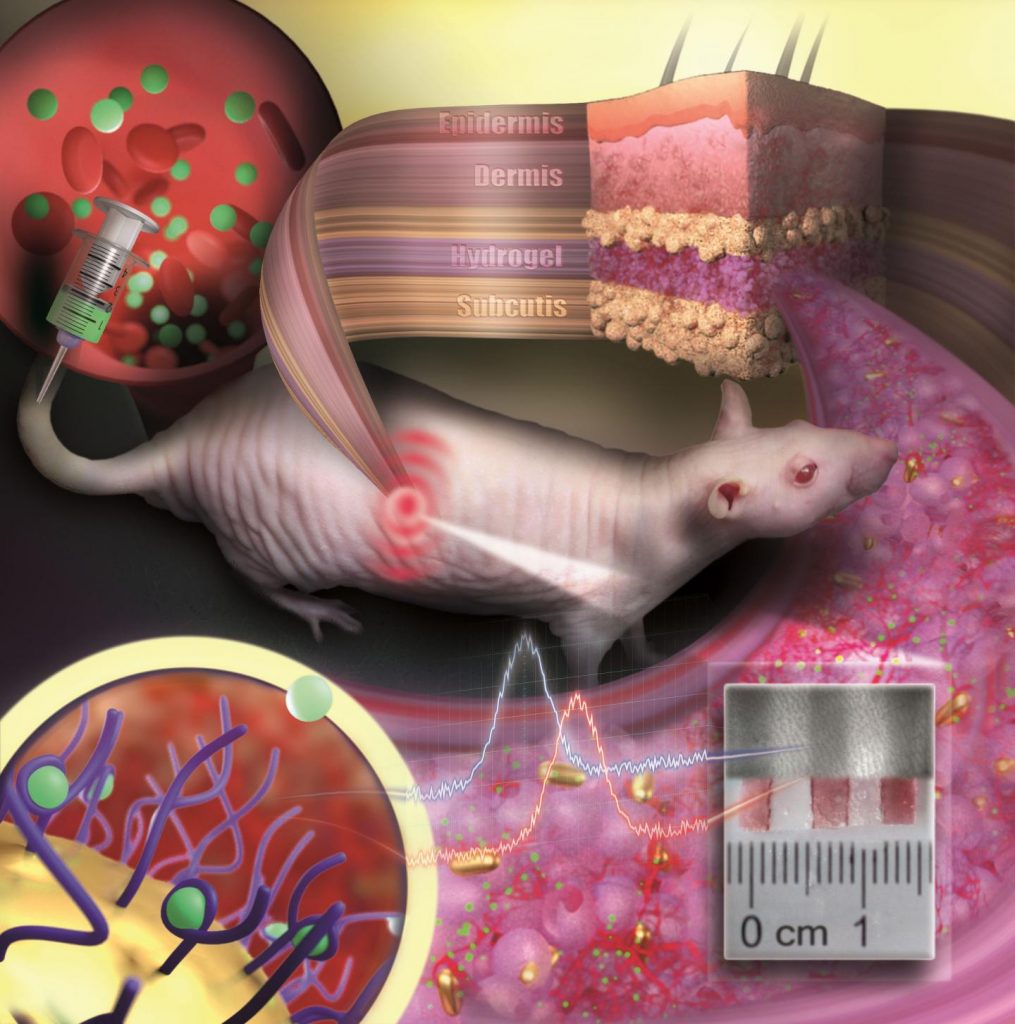
Using this substance [hyaluronic acid], an injectable hydrogel was developed for “tissue prostheses”, which can temporarily fill the gap of the missing muscle/nerve tissues while it regenerates. The injectable nature of this material gives it a significant advantage over traditional bioelectronic devices, which are unsuitable for narrow, deep, or small areas, and necessitate invasive surgeries.
Thanks to its highly “tissue-like” properties, this hydrogel seamlessly interfaces with biological tissues and can be easily administered to hard-to-reach body areas without surgery. The reversible and irreversible crosslinks within the hydrogel adapt to high shear stress during injection, ensuring excellent mechanical stability. This hydrogel also incorporates gold nanoparticles, which gives it decent electrical properties. Its conductive nature allows for the effective transmission of electrophysiological signals between the two ends of injured tissues. In addition, the hydrogel is biodegrdable, meaning that the patients do not need to get surgery again.
With mechanical properties akin to natural tissues, exceptional tissue adhesion, and injectable characteristics, researchers believe this material offers a novel approach to rehabilitation.
Next, the researchers put this novel idea to the test in rodent models. To simulate volumetric muscle loss injury, a large chunk of muscle has been removed from the hind legs of these animals. By injecting the hydrogel and implanting the two kinds of stretchable tissue-interfacing devices for electrical sensing and stimulation, the researchers were able to improve the gait in the “injured” rodents. The hydrogel prosthetics were combined with robot assistance, guided by muscle electromyography signals. Together, the two helped enhance the animal’s gait without nerve stimulation. Furthermore, muscle tissue regeneration was effectively improved over the long term after the conductive hydrogel was used to fill muscle damage.
The injectable conductive hydrogel developed in this study excels in electrophysiological signal recording and stimulation performance, offering the potential to expand its applications. It presents a fresh approach to the field of bioelectronic devices and holds promise as a soft tissue prosthesis for rehabilitation support.
Emphasizing the significance of the research, Professor SHIN Mikyung notes, “We’ve created an injectable, mechanically tough, and electrically conductive soft tissue prosthesis ideal for addressing severe muscle damage requiring neuromusculoskeletal rehabilitation. The development of this injectable hydrogel, utilizing a novel cross-linking method, is a notable achievement. We believe it will be applicable not only in muscles and peripheral nerves but also in various organs like the brain and heart.”
Professor SON Donghee added, “In this study, the closed-loop gait rehabilitation system entailing tough injectable hydrogel and stretchable and self-healing sensors could significantly enhance the rehabilitation prospects for patients with neurological and musculoskeletal challenges. It could also play a vital role in precise diagnosis and treatment across various organs in the human body.”
The research team is currently pursuing further studies to develop new materials for nerve and muscle tissue regeneration that can be implanted in a minimally invasive manner. They are also exploring the potential for recovery in various tissue damages through the injection of the conductive hydrogel, eliminating the need for open surgery.
Here’s a link to and a citation for the paper,
Injectable tissue prosthesis for instantaneous closed-loop rehabilitation by Subin Jin, Heewon Choi, Duhwan Seong, Chang-Lim You, Jong-Sun Kang, Seunghyok Rho, Won Bo Lee, Donghee Son & Mikyung Shin. Nature volume 623, pages 58–65 (2023) DOI: https://doi.org/10.1038/s41586-023-06628-x Published: 01 November 2023 Issue Date: 02 November 2023
This paper is behind a paywall.