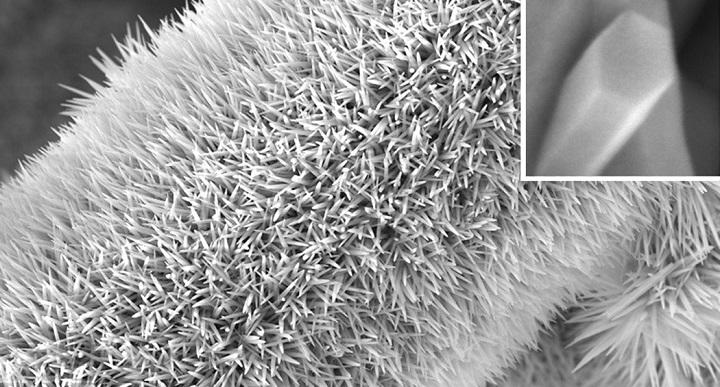
Coincidentally or not, this research from Australia was announced a little more than a month after reports of a major oil spill in the Russian Arctic. A July 10, 2020 news item on phys.org announces a new technology for mopping up oil spills (Note: Links have been removed),
Oil spill disasters on land cause long-term damage for communities and the natural environment, polluting soils and sediments and contaminating groundwater.
Current methods using synthetic sorbent materials can be effective for cleaning up oil spills, but these materials are often expensive and generate large volumes of non-biodegradable plastic wastes. Now the first comparison of natural-origin sorbent materials for land-based oil spills, including peat moss, recycled human hair, and dog fur, shows that sustainable, cheaper and biodegradable options can be developed.
The University of Technology Sydney (UTS) project found that dog fur and human hair products—recycled from salon wastes and dog groomers—can be just as good as synthetic fabrics at cleaning up crude oil spills on hard land surfaces like highway roads, pavement, and sealed concrete floors. Polypropylene, a plastic, is a widely-used fabric used to clean up oil spills in aquatic environments.
…
A July 9, 2020 Univesity of Technology Sydney press release on EurekAlert completes the story,
“Dog fur in particular was surprisingly good at oil spill clean-up, and felted mats from human hair and fur were very easy to apply and remove from the spills.” lead author of the study, UTS Environmental Scientist Dr Megan Murray, said. Dr Murray investigates environmentally-friendly solutions for contamination and leads The Phyto Lab research group at UTS School of Life Sciences.
“This is a very exciting finding for land managers who respond to spilled oil from trucks, storage tanks, or leaking oil pipelines. All of these land scenarios can be treated effectively with sustainable-origin sorbents,” she said.
The sorbents tested included two commercially-available products, propylene and loose peat moss, as well as sustainable-origin prototypes including felted mats made of dog fur and human hair. Prototype oil-spill sorbent booms filled with dog fur and human hair were also tested. Crude oil was used to replicate an oil spill. The results of the study are published in Environments.
The research team simulated three types of land surfaces; non-porous hard surfaces, semi-porous surfaces, and sand, to recreate common oil-spill scenarios.
“We found that loose peat moss is not as effective at cleaning up oil spills on land compared to dog fur and hair products, and it is not useful at all for sandy environments.” Dr Murray said.
“Based on this research, we recommend peat moss is no longer used for this purpose. Given that peat moss is a limited resource and harvesting it requires degrading wetland ecosystems, we think this is a very important finding.” she said.
The research concluded that, for now, sandy environments like coastal beaches can still benefit from the use of polypropylene sorbents, but further exploration of sustainable-origin sorbents is planned.
The researchers say that future applications from the research include investigating felted mats of sustainable-origin sorbents for river bank stabilisation, [emphases mine] as well as the removal of pollutants from flowing polluted waters, similar to existing membrane technology.
Here’s a link to and a citation for the study,
Decontaminating Terrestrial Oil Spills: A Comparative Assessment of Dog Fur, Human Hair, Peat Moss and Polypropylene Sorbents by Megan L. Murray, Soeren M. Poulsen and Brad R. Murray. Environments 2020, 7(7), 52; DOI: https://doi.org/10.3390/environments7070052 Published: 8 July 2020 (This article belongs to the Special Issue Pollution Prevention/Environmental Sustainability for Industry)
This paper is open access.
As for the Russian oil spill
A June 4, 2020 British Broadcasting Corporation (BBC) news online article outlines the situation regarding the oil spill and the steps being taken to deal with it,
Russia’s President Vladimir Putin has declared a state of emergency after 20,000 tonnes of diesel oil leaked into a river within the Arctic Circle.
The spill happened when a fuel tank at a power plant near the Siberian city of Norilsk collapsed last Friday [May 29, 2020].
The power plant’s director Vyacheslav Starostin has been taken into custody until 31 July, but not yet charged.
The plant is owned by a subsidiary of Norilsk Nickel, which is the world’s leading nickel and palladium producer.
The Russian Investigative Committee (SK) has launched a criminal case over the pollution and alleged negligence, as there was reportedly a two-day delay in informing the Moscow authorities about the spill.
Ground subsidence beneath the fuel storage tanks is believed to have caused the spill. Arctic permafrost has been melting in exceptionally warm weather [more information about the weather towards the end of this posting] for this time of year.
…
Russian Minister for Emergencies Yevgeny Zinichev told Mr Putin that the Norilsk plant had spent two days trying to contain the spill, before alerting his ministry.
The leaked oil drifted some 12km (7.5 miles) from the accident site, turning long stretches of the Ambarnaya river crimson red.

Getting back to the June 4, 2020 British Broadcasting Corporation (BBC) news online article,
“Why did government agencies only find out about this two days [May 29, 2020?) after the fact?” he asked the subsidiary’s chief, Sergei Lipin. “Are we going to learn about emergency situations from social media?”
The region’s governor, Alexander Uss, had earlier told President Putin that he became aware of the oil spill on Sunday [May 31, 2020] after “alarming information appeared in social media”.
The spill has contaminated a 350 sq km (135 sq mile) area, state media report.
…
The state of emergency means extra forces are going to the area to assist with the clean-up operation.
The accident is believed to be the second largest in modern Russian history in terms of volume, an expert from the World Wildlife Fund, Alexei Knizhnikov, told the AFP [Agence France Presse] news agency.
The incident has prompted stark warnings from environmental groups, who say the scale of the spill and geography of the river mean it will be difficult to clean up.
Greenpeace has compared it to the 1989 Exxon Valdez disaster in Alaska.
Oleg Mitvol, former deputy head of Russia’s environmental watchdog Rosprirodnadzor, said there had “never been such an accident in the Arctic zone”.
He said the clean-up could cost 100bn roubles (£1.2bn; $1.5bn) and take between five and 10 years.
Minister of Natural Resources Dmitry Kobylkin warned against trying to burn off such a vast quantity of fuel oil.
He proposed trying to dilute the oil with reagents. Only the emergencies ministry with military support could deal with the pollution, he said.
Barges with booms could not contain the slick because the Ambarnaya river was too shallow, he warned.
He suggested pumping the oil on to the adjacent tundra, although President Putin added: “The soil there is probably saturated [with oil] already.”
An update of the situation can be found in a July 8, 2020 Canadian Broadcasting Corporation (CBC) article (issued by Thomson Reuters),
Russia’s environmental watchdog has asked a power subsidiary of Russian mining giant Norilsk Nickel to pay almost 148 billion rubles, or $2.8 billion Cdn, in damages over an Arctic fuel spill in Siberia.
Rosprirodnadzor, the Federal Service for Supervision of Use of Natural Resources, said in a statement on Monday [July 8, 2020] that it had already sent a request for “voluntary compensation” to the subsidiary, NTEK, after calculating the damage caused by the May 29 [2020] fuel spill.
Norilsk Nickel’s Moscow-listed shares fell by 3 per cent after the watchdog’s statement.
A fuel tank at the power plant lost pressure and released 21,000 tonnes of diesel into rivers and subsoil near the city of Norilsk, 2,900 kilometres northeast of Moscow. Russian President Vladimir Putin subsequently declared a state of emergency in the region, and investigators detained three staff at the power plant.
…
Norilsk, a remote city of 180,000 people situated 300 kilometres inside the Arctic Circle, is built around Norilsk Nickel, the world’s leading nickel and palladium producer, and has a reputation for its pollution.
Rosprirodnadzor said the damages included the cost for nearby water bodies, estimated at 147.05 billion rubles, $2.8 billion Cdn, and for subsoil, estimated at 738.62 million roubles, $14 million Cdn.
…
I can’t find any August 2020 updates for the oil spill situation in Russia. (Note: There is now an oil spill in a ecologically sensitive region near Mauritius; see August 13, 2020 news item on CBC news online website.)
Exceptionally warm weather
The oil spill isn’t the only problem in the Arctic.Here’s more from a June 23, 2020 article by Matt Simon for Wired magazine (Note: A link has been removed),
On Saturday [June 20, 2020], the residents of Verkhoyansk, Russia, marked the first day of summer with 100 degree Fahrenheit temperatures. Not that they could enjoy it, really, as Verkhoyansk is in Siberia, hundreds of miles from the nearest beach. That’s much, much hotter than towns inside the Arctic Circle usually get. That 100 degrees appears to be a record, well above the average June high temperature of 68 degrees. Yet it’s likely the people of Verkhoyansk will see that record broken again in their lifetimes: The Arctic is warming twice as fast as the rest of the planet—if not faster—creating ecological chaos for the plants and animals that populate the north.
“The events over the weekend—in the last few weeks, really—with the heatwave in Siberia, all are unprecedented in terms of the magnitude of the extremes in temperature,” says Sophie Wilkinson, a wildfire scientist at McMaster University who studies northern peat fires, which themselves have grown unusually frequent in recent years as temperatures climb.
The Arctic’s extreme warming, known as Arctic amplification or polar amplification, may be due to three factors. One, the region’s reflectivity, or albedo—how much light it bounces back into space—is changing as the world warms. “What we’ve been seeing over the last 30 years is some relatively dramatic declines in sea ice in the summertime,” says University of Edinburgh global change ecologist Isla Myers-Smith, who studies the Arctic.
Since ice is white, it reflects the sun’s energy, something you’re already probably familiar with when it comes to staying cool in the summer. If you had to pick the color of T-shirt to wear when going hiking on a hot day, she says, “most of us would pick the white T-shirt, because that’s going to reflect the sun’s heat off of our back.” Similarly, Myers-Smith says, “If the sea ice melts in the Arctic, that will remove that white surface off of the ocean, and what will be exposed is this darker ocean surface that will absorb more of the sun’s heat.”
…
If you’re interested in the environmental consequences of the warming of the Arctic, this is a very good article.
Finishing up, I wish the clean-up crews (in Russia and near Mauritius) all the best as they work in the midst of a pandemic, as well as, an environmental disaster (both the oil spill and the warming of the Arctic).