This is the middle commentary on the report titled,(INVESTING IN CANADA’S FUTURE; Strengthening the Foundations of Canadian Research). Part 1 of my commentary having provided some introductory material and first thoughts about the report, this part offers more detailed thoughts and Part 3 offers ‘special cases’ and sums up some of the ideas first introduced in part 1.
The report: the good, the informative, and the problematic
As Canadian government reports go, this is quite readable and I’m delighted to note some sections are downright engaging. (Thank you to the writer)
Happily, the report acknowledges the problems with the usual measures for research performance (p. xiv print; p. 18 PDF in the Executive Summary and, also, in Chapter 3). Also happily, the panel describes how the scope of the disciplines was decided,
Among the early challenges for the Panel were misinterpretation of its moniker and the related scope of its work. The term “fundamental science” originated with federal Budget 2016, which announced the Government of Canada’s intent to undertake a review.3 Alignment of terminology followed. Some members of the anglophone research community were understandably concerned that the Panel’s mandate excluded applied science in a range of fields, as well as the social sciences and humanities. Francophone researchers, accustomed to les sciences sociales et humaines, were more sanguine.
Minister Duncan [Kirsty Duncan], whose own scholarship cuts across the natural sciences, social sciences, and humanities, made it clear from the outset that the Panel was to examine the full range of scientific and scholarly disciplines. The Panel’s secretariat and members similarly emphasized the breadth of our review. We were accordingly delighted to receive submissions from many researchers and organizations representative of disciplines supported by the three granting councils, others doing transdisciplinary research who sometimes find themselves in limbo, and others again frustrated that the lack of collaboration across the councils has effectively shut out their disciplines altogether.
A residual source of some confusion was the term “fundamental”, which is used infrequently in the social sciences and humanities even though much scholarship in those fields is arguably basic or conceptual.
The Panel again took a pragmatic view. Our mandate was derived in meaningful measure from concerns that Canada’s capacity for generation of exciting new knowledge had been eroded. We therefore assumed our remit ranged from basic science focused on making major discoveries to applied science with important technological implications, and from deep philosophical inquiry to rigorous economic evaluations of policies and programs.
The Panel emphasizes in this latter regard that societies without great science and scholarship across a wide range of disciplines are impoverished in multiple dimensions. From the social sciences and humanities, contributions range from deeper understanding of the complexity of human nature and social structures to grace in self-expression and excellence and beauty in the creative and performing arts. From the natural and health sciences and engineering, while attention often focuses on practical applications, basic research provides the breakthrough insights that fundamentally change our understanding of the natural world and our cosmos. We return to this subject in Chapter 2.
The Panel also observes that these categorizations are all focused on research subject matter, when in fact the subject that really matters may be the person doing the research. Postsecondary education enriched by exposure to basic research provides citizens with an outlook and intellectual tools that are extraordinarily well-suited to technological and social innovation. Indeed, countless authors of abstract graduate theses have gone on to lives of deep and productive engagement with practical problems, bringing with them perspectives that reflect an inquiring and critical mind.
In brief, the Panel’s primary interest is in the extramural research realm, and particularly in supports for research into topics chosen by scholars and scientists from the full range of disciplines, using methods that they have developed or adapted, and subject to review by research colleagues. This research may be basic or applied. It may be project-based or programmatic. And it may have early application or no immediate relevance. However, a key criterion is that the work is sufficiently excellent to withstand critical scrutiny by peers, [emphasis mine] and produces knowledge that, after appropriate review, can be shared widely to advance the collective store of knowledge and ideas in the relevant field or fields. (p. 4-5 print; pp. 38-9 PDF)
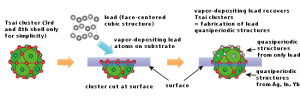
Here’s a problem not mentioned in the report. Sometimes, the most exciting work is not appreciated or even approved by your peers. Daniel Schechtman’s work with quasicrystals illustrates the issue (from the Dan Schechtman Wikipedia entry),
“I was a subject of ridicule and lectures about the basics of crystallography. The leader of the opposition to my findings was the two-time Nobel Laureate Linus Pauling, [emphasis mine] the idol of the American Chemical Society and one of the most famous scientists in the world. For years, ’til his last day, he fought against quasi-periodicity in crystals. He was wrong, and after a while, I enjoyed every moment of this scientific battle, knowing that he was wrong.”[citation needed]
Linus Pauling is noted saying “There is no such thing as quasicrystals, only quasi-scientists.”[15] Pauling was apparently unaware of a paper in 1981 by H. Kleinert and K. Maki which had pointed out the possibility of a non-periodic Icosahedral Phase in quasicrystals[16] (see the historical notes). The head of Shechtman’s research group told him to “go back and read the textbook” and a couple of days later “asked him to leave for ‘bringing disgrace’ on the team.”[17] [emphasis mine] Shechtman felt dejected.[15] On publication of his paper, other scientists began to confirm and accept empirical findings of the existence of quasicrystals.[18][19]
Schechtman does get back into the lab, finds support for his discovery from other scientists, and wins the Nobel Prize for Chemisty in 2011. But, that first few years was pretty rough sledding. As for the problem, how can you tell the difference between ground-breaking research and a ‘nutbar’ theory?
Getting back to the report, there’s a very nice listing of research milestones (the inception of various funding agencies, science ministries, important reports, and more) in the Canadian research landscape on pp. 8-9 print; pp. 42-3 PDF. The list stretches from 1916 to 2016. Oddly, the 2011 Jenkins report (also known as the Review of Federal Support to R&D report) is not on the list. Of course, it was a report commissioned by the then Conservative federal government.
Chapter 2 is the ‘Case for Science and Inquiry’ and it includes a bit of a history of the world, geologically speaking (p. 18 print; p. 52 PDF), and more. The scholars that are referenced tend to be from Europe and the US (sigh … isn’t there a way to broaden our perspectives?).
I was surprised that they didn’t include Wilder Penfield’s work in their partial listing of Canadian discoveries, and achievements in natural sciences, engineering, and health (p. 22 print; p. 56 PDF). From the Wilder Penfield Wikipedia entry*,
Wilder Graves Penfield OM CC CMG FRS[1] (January 26, 1891 – April 5, 1976) was an American-Canadian pioneering neurosurgeon once dubbed “the greatest living Canadian.”[2] He expanded brain surgery’s methods and techniques, including mapping the functions of various regions of the brain such as the cortical homunculus. His scientific contributions on neural stimulation expand across a variety of topics including hallucinations, illusions, and déjà vu. Penfield devoted a lot of his thinking to mental processes, including contemplation of whether there was any scientific basis for the existence of the human soul.[2]
Also mildly surprising was Ursula Franklin’s exclusion from their sampling of great Canadian thinkers in the social science and humanities (p. 23 print; p. 57 PDF) especially as there seems to be room for one more entry. From the Ursula Franklin Wikipedia entry,
Ursula Martius Franklin, CC OOnt FRSC (16 September 1921 – 22 July 2016), was a German-Canadian metallurgist, research physicist, author, and educator who taught at the University of Toronto for more than 40 years.[1] …
Franklin is best known for her writings on the political and social effects of technology. For her, technology was much more than machines, gadgets or electronic transmitters. It was a comprehensive system that includes methods, procedures, organization, “and most of all, a mindset”.[5] …
For some, Franklin belongs in the intellectual tradition of Harold Innis and Jacques Ellul who warn about technology’s tendency to suppress freedom and endanger civilization.[8] …
As noted earlier, Chapter 3 offers information about typical measures for scientific impact. There were two I didn’t mention. First, there are the scores for interprovincial collaboration. While we definitely could improve our international collaboration efforts, it’s the interprovincial efforts that tend to be pitiful (Note: I’ve had to create the table myself so it’s not identical to the report table’s format),
| Province or Territory |
Collaborative rates 2003-2014 |
| Interprovincial |
International |
| Alberta |
24.4 |
42.5 |
| British Columbia |
23.0 |
48.2 |
| Manitoba |
33.5 |
39.7 |
| New Brunswick |
35.7 |
38.0 |
| Newfoundland and Labrador |
33.6 |
38.7 |
| Northwest Territories |
86.9 |
32.5 |
| Nova Scotia |
34.7 |
40.9 |
| Nunavut |
85.7 |
34.5 |
| Ontario |
14.8 |
43.4 |
| Prince Edward island |
46.7 |
40.6 |
| Québec |
16.9 |
43.8 |
| Saskatchewan |
33.9 |
41.7 |
| Yukon |
79.4 |
39.0 |
| Canada |
9.8 |
43.7 |
* *The interprovincial collaboration rates (IPC) are computed on whole counts, not fractional counts. So, for example, a publication with authors from four provinces would count as one for Canada and one for each of the provinces. So the IPC for the whole of Canada would be 1 out of 874,475 (Canada’s whole publication count over 2003–2014) and the IPC for Ontario (for example) would be 1 out of 396,811 (the whole count for Ontario). Therefore the interprovincial collaboration rate would be lower for Canada than for Ontario. (p. 39 print; 73 PDF)
Second, there are the prizes,
Moving from highly-cited researchers and papers to the realm of major international research prizes takes us further into the realm of outlying talent. Major international prizes for research are relevant measures because they bring great prestige not just to individuals and teams, but also to institutions and nations. They are also the culmination of years of excellence in research and, particularly when prizes are won repeatedly across a range of disciplines, they send strong signals to the world about the health of a nation’s basic research ecosystem.
Unfortunately, Canada’s performance in winning international prizes is also lagging. In 2013 the Right Honourable David Johnston, Governor General of Canada, and Dr Howard Alper, then chair of the national Science, Technology and Innovation Council (STIC), observed that Canadians underperform “when it comes to the world’s most distinguished awards”, e.g., Nobel Prize, Wolf Prize, and Fields Medal. They added: “In the period from 1941 to 2008, Canadians received 19 of the top international awards in science—an impressive achievement, to be sure, but lacking when compared with the United States (with 1,403 winners), the United Kingdom (222), France (91), Germany (75) and Australia (42).”22 ix
There is an interesting wrinkle to the dominance of the U.S. in Nobel prizes.23 Over 30 per cent of all U.S. Nobel laureates since 1950 were foreign-born, with that proportion rising over time. From 2007 to 2016, the 54 Nobel prizes awarded to U.S.-based researchers included 20 immigrants. Sources differ as to whether more of the U.S. Nobel laureates originated from Canada or Germany, but the best estimate is that, since 1901, there have been 15 Canadian-born, and in many cases Canadian-educated, Nobel laureates based in the U.S.—double the total number of Nobel prizes awarded to Canadian-based researchers in the same period.
From the standpoint of international recognition, 2015 was an exceptional year. Canadians won two of the pinnacle awards: a Nobel prize (Arthur McDonald for Physics) and a Wolf prize (James Arthur for Mathematics). Those prizes celebrate work that exemplifies two very different models of discovery. As a theoretical mathematician, Dr Arthur’s pioneering papers in automorphic forms have been overwhelmingly sole-authored; his long-term support has come from modest NSERC Discovery Grants. As a particle physicist, Dr McDonald has led a large team in developing and operating the renowned Sudbury Neutrino Laboratory, a major science facility purpose-built deep in an active nickel mine, where startling observations have been made that are forcing a reconsideration of The Standard Model for Elementary Particles. In both cases, however, what matters is that the work began decades ago, and Canada provided long-term support at the levels and in forms required to enable path-breaking discoveries to be made.
Canada cannot assume that there will be a series of other pinnacle prizes awarded based on discoveries that tap into work initiated in the 1970s and 1980s. To ensure a continuous pipeline of successful nominations for international awards, research institutions must be supported consistently to recruit and retain outstanding scholars and scientists. They in turn must be supported to create world-class research environments through meritocratic adjudication processes that offer fair access to appropriate levels of consistent funding for scientific inquiry. Our assessment thus far has not given us great confidence that these winning conditions are being created, let alone enhanced. (pp. 46-7 print; pp. 80-1 PDF)
I found one more interesting bit in the report, a dated list of Canadian science advice vehicles. Somewhat optimistically given the speed with which the initiative has moved forward, they’ve listed a Canadian chief science advisor for 2017 (p. 54 print; p. 88 PDF). Understandably, since it is a recommendation, they left out the NACRI, .
Again, here’s a link to the other parts:
INVESTING IN CANADA’S FUTURE; Strengthening the Foundations of Canadian Research (Review of fundamental research final report) Commentaries
Part 1
Part 3
*’enty’ corrected to ‘entry’ and a link to Wilder Penfield’s Wikipedia entry was added on June 15, 2017.