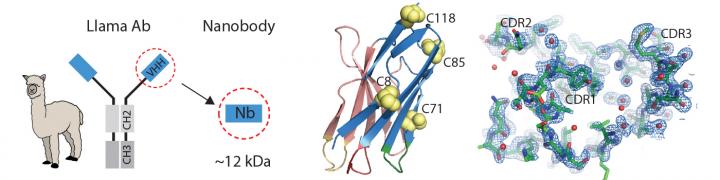
Bryson and Sanchez are not the first camelids to grace this blog. ‘Llam’ me lend you some antibodies—antibody particles extracted from camels and llamas, a June 12, 2014 posting, and Llama-derived nanobodies are good for solving crystal structure, a December 14, 2017 posting, both feature news about medical breakthroughs with regard to the antibodies found in Llamas, camels, and other camelids (including alpacas) could enable.
The latest camelid-oriented medical research story is in an April 11, 2019 news item on phys.org (Note: A link has been removed),
In 1989, two undergraduate students at the Free University of Brussels were asked to test frozen blood serum from camels, and stumbled on a previously unknown kind of antibody. It was a miniaturized version of a human antibody, made up only of two heavy protein chains, rather than two light and two heavy chains. As they eventually reported, the antibodies’ presence was confirmed not only in camels, but also in llamas and alpacas.
Fast forward 30 years. In the journal PNAS [Proceedings of the National Academy of Science] this week [April 8 – 12, 2019], researchers at Boston Children’s Hospital and MIT [Massachusetts Institute of Technology] show that these mini-antibodies, shrunk further to create so-called nanobodies, may help solve a problem in the cancer field: making CAR T-cell therapies work in solid tumors.
…
An April 11, 2019 Boston Children’s Hospital news release on EurekAlert, which originated the news item, explores the technology,
Highly promising for blood cancers, chimeric antigen receptor (CAR) T-cell therapy genetically engineers a patient’s own T cells to make them better at attacking cancer cells. The Dana-Farber/Boston Children’s Cancer and Blood Disorders Center is currently using CAR T-cell therapy for relapsed acute lymphocytic leukemia (ALL), for example.
But CAR T cells haven’t been good at eliminating solid tumors. It’s been hard to find cancer-specific proteins on solid tumors that could serve as safe targets. Solid tumors are also protected by an extracellular matrix, a supportive web of proteins that acts as a barrier, as well as immunosuppressive molecules that weaken the T-cell attack.
Rethinking CAR T cells
That’s where nanobodies come in. For two decades, they largely remained in the hands of the Belgian team. But that changed after the patent expired in 2013. [emphases mine]
“A lot of people got into the game and began to appreciate nanobodies’ unique properties,” says Hidde Ploegh, PhD, an immunologist in the Program in Cellular and Molecular Medicine at Boston Children’s and senior investigator on the PNAS study.
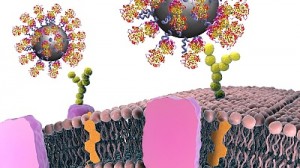
One useful attribute is their enhanced targeting abilities. Ploegh and his team at Boston Children’s, in collaboration with Noo Jalikhani, PhD, and Richard Hynes, PhD at MIT’s Koch Institute for Integrative Cancer Research, have harnessed nanobodies to carry imaging agents, allowing precise visualization of metastatic cancers.
The Hynes team targeted the nanobodies to the tumors’ extracellular matrix, or ECM — aiming imaging agents not at the cancer cells themselves, but at the environment that surrounds them. Such markers are common to many tumors, but don’t typically appear on normal cells.
“Our lab and the Hynes lab are among the few actively pursuing this approach of targeting the tumor micro-environment,” says Ploegh. “Most labs are looking for tumor-specific antigens.”
Targeting tumor protectors
Ploegh’s lab took this idea to CAR T-cell therapy. His team, including members of the Hynes lab, took aim at the very factors that make solid tumors difficult to treat.
The CAR T cells they created were studded with nanobodies that recognize specific proteins in the tumor environment, bearing signals directing them to kill any cell they bound to. One protein, EIIIB, a variant of fibronectin, is found only on newly formed blood vessels that supply tumors with nutrients. Another, PD-L1, is an immunosuppressive protein that most cancers use to silence approaching T cells.
Biochemist Jessica Ingram, PhD of the Dana-Farber Cancer Institute, Ploegh’s partner and a coauthor on the paper, led the manufacturing pipeline. She would drive to Amherst, Mass., to gather T cells from two alpacas, Bryson and Sanchez, inject them with the antigen of interest and harvest their blood for further processing back in Boston to generate mini-antibodies.
Taking down melanoma and colon cancer
Tested in two separate melanoma mouse models, as well as a colon adenocarcinoma model in mice, the nanobody-based CAR T cells killed tumor cells, significantly slowed tumor growth and improved the animals’ survival, with no readily apparent side effects.
Ploegh thinks that the engineered T cells work through a combination of factors. They caused damage to tumor tissue, which tends to stimulate inflammatory immune responses. Targeting EIIIB may damage blood vessels in a way that decreases blood supply to tumors, while making them more permeable to cancer drugs.
“If you destroy the local blood supply and cause vascular leakage, you could perhaps improve the delivery of other things that might have a harder time getting in,” says Ploegh. “I think we should look at this as part of a combination therapy.”
Future directions
Ploegh thinks his team’s approach could be useful in many solid tumors. He’s particularly interested in testing nanobody-based CAR T cells in models of pancreatic cancer and cholangiocarcinoma, a bile duct cancer from which Ingram passed away in 2018.
The technology itself can be pushed even further, says Ploegh.
“Nanobodies could potentially carry a cytokine to boost the immune response to the tumor, toxic molecules that kill tumor and radioisotopes to irradiate the tumor at close range,” he says. “CAR T cells are the battering ram that would come in to open the door; the other elements would finish the job. In theory, you could equip a single T cell with multiple chimeric antigen receptors and achieve even more precision. That’s something we would like to pursue.”
So, the Belgian researchers have a patent for two decades and, after it expires, more researchers could help to take the work further. Hmm …
Moving on, here’s a link to and a citation for the paper,
Nanobody-based CAR T cells that target the tumor microenvironment inhibit the growth of solid tumors in immunocompetent mice by Yushu Joy Xie, Michael Dougan, Noor Jailkhani, Jessica Ingram, Tao Fang, Laura Kummer, Noor Momin, Novalia Pishesha, Steffen Rickelt, Richard O. Hynes, and Hidde Ploegh. PNAS DOI: https://doi.org/10.1073/pnas.1817147116
First published April 1, 2019
This paper is behind a paywall