The Argus II artificial retina was first mentioned here in a Feb. 15, 2013 posting (scroll down about 50% of the way) when it received US Food and Drug Administration (FDA) commercial approval. In retrospect that seems puzzling since the results of a three-year clinical trial have just been reported in a June 23, 2015 news item on ScienceDaily (Note: There was one piece of information about the approval which didn’t make its way into the information disseminated in 2013),
The three-year clinical trial results of the retinal implant popularly known as the “bionic eye,” have proven the long-term efficacy, safety and reliability of the device that restores vision in those blinded by a rare, degenerative eye disease. The findings show that the Argus II significantly improves visual function and quality of life for people blinded by retinitis pigmentosa. They are being published online in Ophthalmology, the journal of the American Academy of Ophthalmology.
A June 23, 2015 American Academy of Ophthalmology news release (also on EurekAlert), which originated the news item, describes the condition the Argus II is designed for and that crucial bit of FDA information,
Retinitis pigmentosa is an incurable disease that affects about 1 in 4,000 Americans and causes slow vision loss that eventually leads to blindness.[1] The Argus II system was designed to help provide patients who have lost their sight due to the disease with some useful vision. Through the device, patients with retinitis pigmentosa are able to see patterns of light that the brain learns to interpret as an image. The system uses a miniature video camera stored in the patient’s glasses to send visual information to a small computerized video processing unit which can be stored in a pocket. This computer turns the image to electronic signals that are sent wirelessly to an electronic device implanted on the retina, the layer of light-sensing cells lining the back of the eye.
The Argus II received Food and Drug Administration (FDA) approval as a Humanitarian Use Device (HUD) in 2013, which is an approval specifically for devices intended to benefit small populations and/or rare conditions. [emphasis mine]
I don’t recall seeing “Humanitarian Use Device (HUD)” in the 2013 materials which focused on the FDA’s commercial use approval. I gather from this experience that commercial use doesn’t necessarily mean they’ve finished with clinical trials and are ready to start selling the product. In any event, I will try to take a closer look at the actual approvals the next time, assuming I can make sense of the language.
After all the talk about it, here’s what the device looks like,
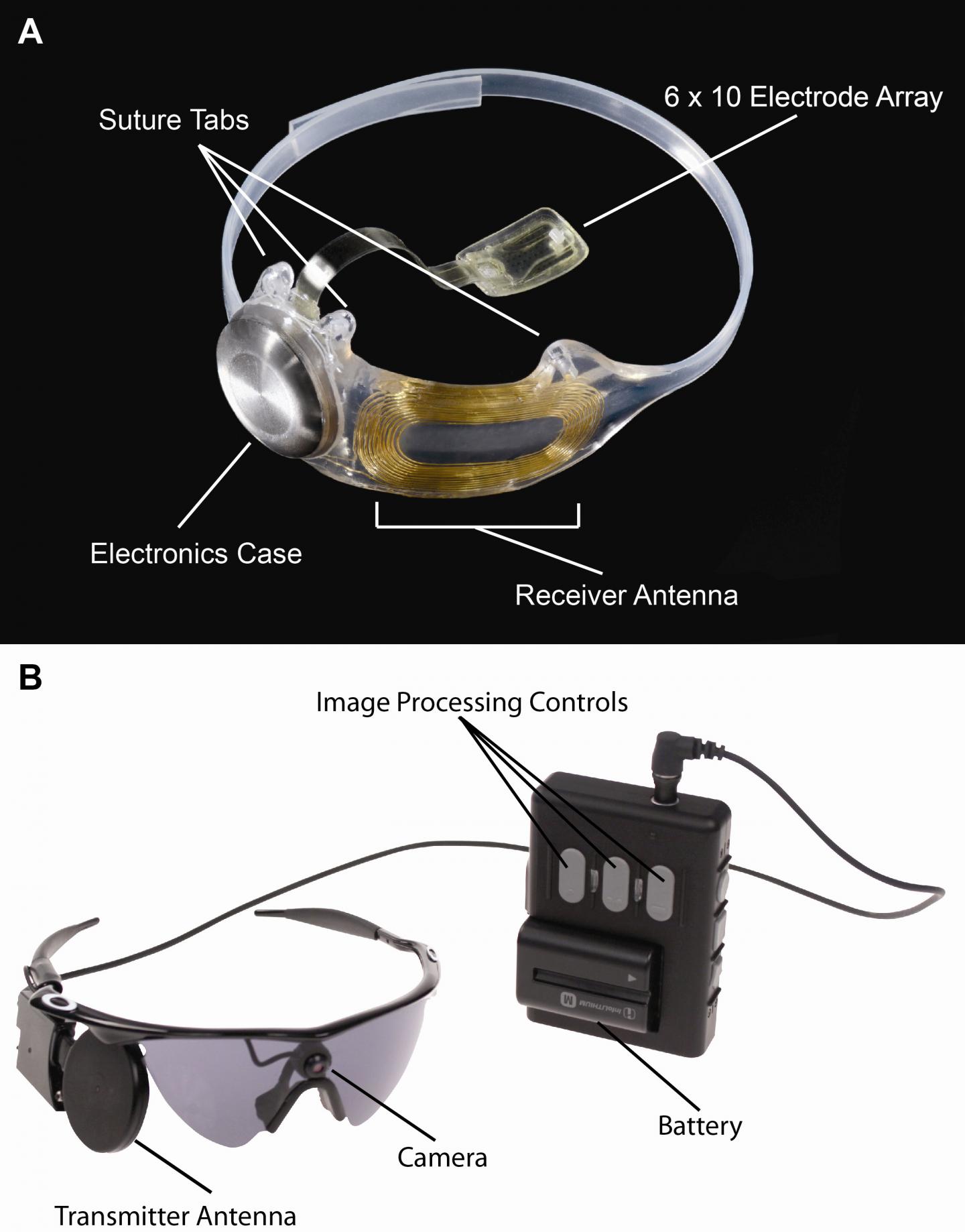
Caption: Figure A, The implanted portions of the Argus II System. Figure B, The external components of the Argus II System. Images in real time are captured by camera mounted on the glasses. The video processing unit down-samples and processes the image, converting it to stimulation patterns. Data and power are sent via radiofrequency link form the transmitter antenna on the glasses to the receiver antenna around the eye. A removable, rechargeable battery powers the system.
Credit: Photo courtesy of Second Sight Medical Products, Inc.
The news release offers more details about the recently completed clinical trial,
To further evaluate the safety, reliability and benefit of the device, a clinical trial of 30 people, aged 28 to 77, was conducted in the United States and Europe. All of the study participants had little or no light perception in both eyes. The researchers conducted visual function tests using both a computer screen and real-world conditions, including finding and touching a door and identifying and following a line on the ground. A Functional Low-vision Observer Rated Assessment (FLORA) was also performed by independent visual rehabilitation experts at the request of the FDA to assess the impact of the Argus II system on the subjects’ everyday lives, including extensive interviews and tasks performed around the home.
The visual function results indicated that up to 89 percent of the subjects performed significantly better with the device. The FLORA found that among the subjects, 80 percent received benefit from the system when considering both functional vision and patient-reported quality of life, and no subjects were affected negatively.
After one year, two-thirds of the subjects had not experienced device- or surgery-related serious adverse events. After three years, there were no device failures. Throughout the three years, 11 subjects experienced serious adverse events, most of which occurred soon after implantation and were successfully treated. One of these treatments, however, was to remove the device due to recurring erosion after the suture tab on the device became damaged.
“This study shows that the Argus II system is a viable treatment option for people profoundly blind due to retinitis pigmentosa – one that can make a meaningful difference in their lives and provides a benefit that can last over time,” said Allen C. Ho, M.D., lead author of the study and director of the clinical retina research unit at Wills Eye Hospital. “I look forward to future studies with this technology which may make possible expansion of the intended use of the device, including treatment for other diseases and eye injuries.”
Here’s a link to a PDF of and a citation for the paper,
Long-Term Results from an Epiretinal Prosthesis to Restore Sight to the Blind by Allen C. Ho,Mark S. Humayun, Jessy D. Dorn, Lyndon da Cruz, Gislin Dagnelie,James Handa, Pierre-Olivier Barale, José-Alain Sahel, Paulo E. Stanga, Farhad Hafezi, Avinoam B. Safran, Joel Salzmann, Arturo Santos, David Birch, Rand Spencer, Artur V. Cideciyan, Eugene de Juan, Jacque L. Duncan, Dean Eliott, Amani Fawzi, Lisa C. Olmos de Koo, Gary C. Brown, Julia A. Haller, Carl D. Regillo, Lucian V. Del Priore, Aries Arditi, Duane R. Geruschat, Robert J. Greenberg. Opthamology, June 2015 http://dx.doi.org/10.1016/j.ophtha.2015.04.032
This paper is open access.