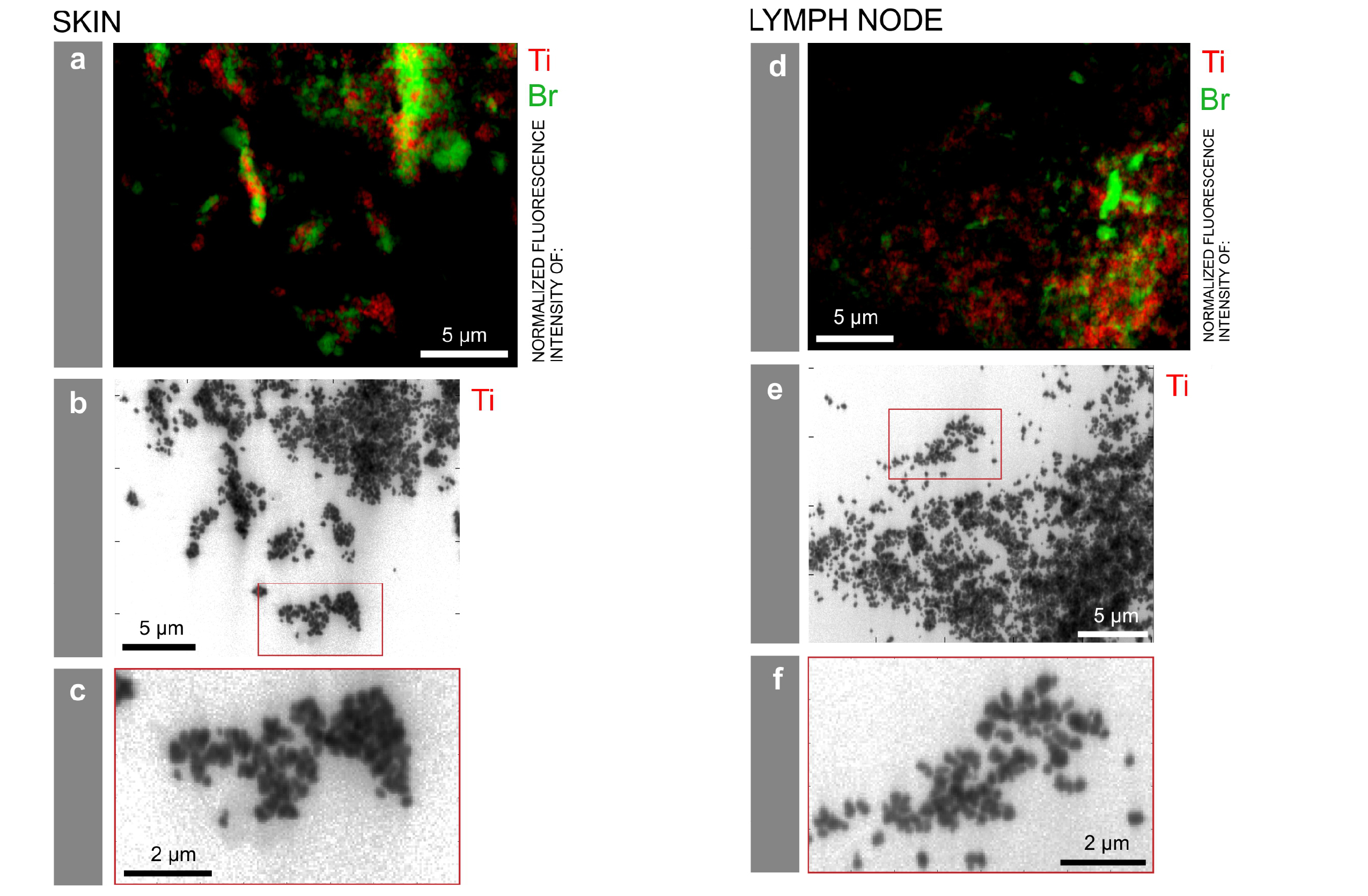
This paper on ethics (aside: I have a few comments after the news release and citation) comes from the US Pacific Northwest National Laboratory (PNNL) according to a July 12, 2023 news item on phys.org,
Prosthetics moved by thoughts. Targeted treatments for aggressive brain cancer. Soldiers with enhanced vision or bionic ears. These powerful technologies sound like science fiction, but they’re becoming possible thanks to nanoparticles.
“In medicine and other biological settings, nanotechnology is amazing and helpful, but it could be harmful if used improperly,” said Pacific Northwest National Laboratory (PNNL) chemist Ashley Bradley, part of a team of researchers who conducted a comprehensive survey of nanobiotechnology applications and policies.
Their research, available in Health Security, works to sum up the very large, active field of nanotechnology in biology applications, draw attention to regulatory gaps, and offer areas for further consideration.
…
A July 12, 2023 PNNL news release (also on EurekAlert), which originated the news item, delves further into the topic, Note: A link has been removed,
“In our research, we learned there aren’t many global regulations yet,” said Bradley. “And we need to create a common set of rules to figure out the ethical boundaries.”
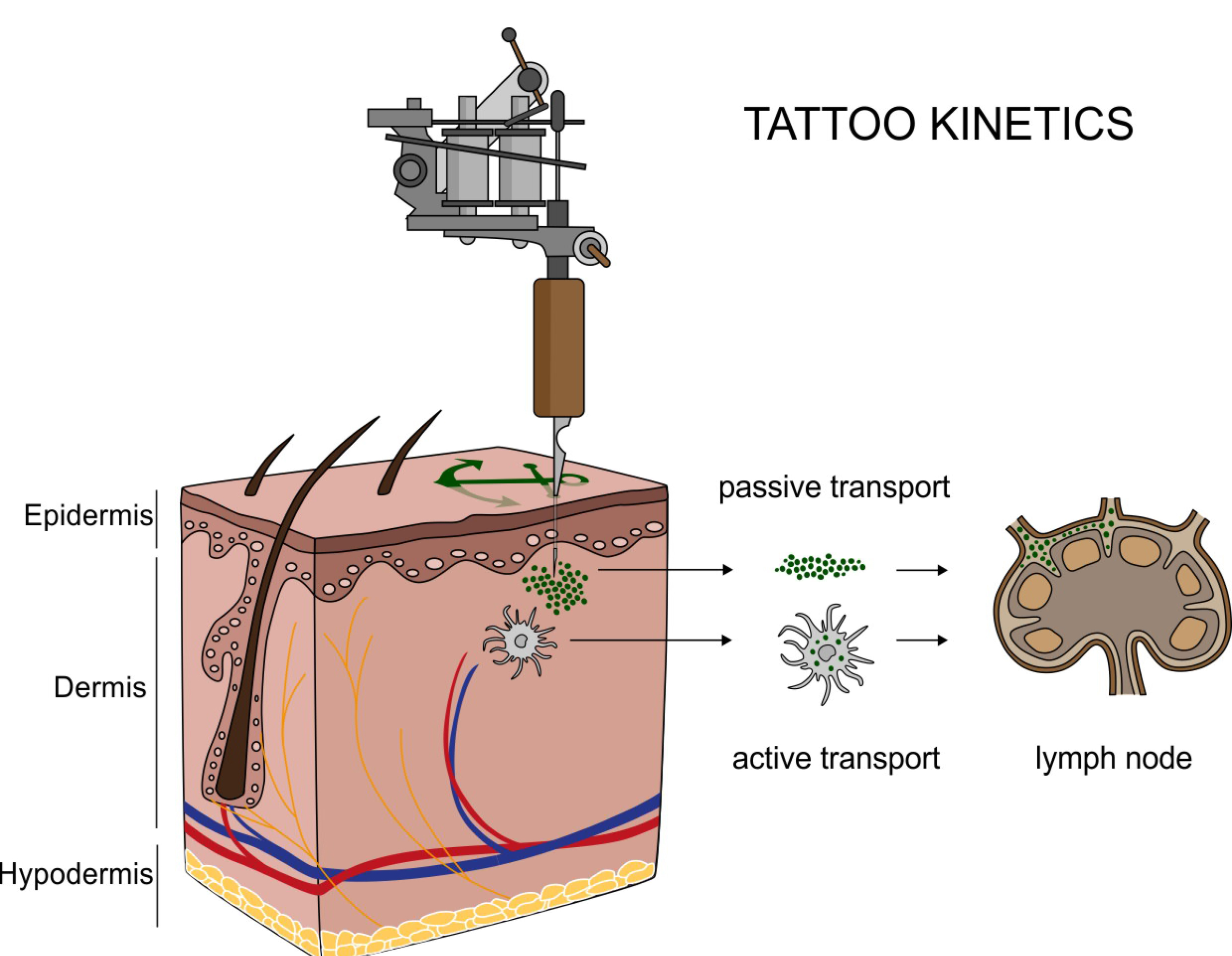
Nanoparticles, big differences
Nanoparticles are clusters of molecules with different properties than large amounts of the same substances. In medicine and other biology applications, these properties allow nanoparticles to act as the packaging that delivers treatments through cell walls and the difficult to cross blood-brain barrier.
“You can think of the nanoparticles a little bit like the plastic around shredded cheese,” said PNNL chemist Kristin Omberg. “It makes it possible to get something perishable directly where you want it, but afterwards you’ve got to deal with a whole lot of substance where it wasn’t before.”
Unfortunately, dealing with nanoparticles in new places isn’t straightforward. Carbon is pencil lead, nano carbon conducts electricity. The same material may have different properties at the nanoscale, but most countries still regulate it the same as bulk material, if the material is regulated at all.
For example, zinc oxide, a material that was stable and unreactive as a pigment in white paint, is now accumulating in oceans when used as nanoparticles in sunscreen, warranting a call to create alternative reef-safe sunscreens. And although fats and lipids aren’t regulated, the researchers suggest which agencies could weigh in on regulations were fats to become after-treatment byproducts.
The article also inventories national and international agencies, organizations, and governing bodies with an interest in understanding how nanoparticles break down or react in a living organism and the environmental life cycle of a nanoparticle. Because nanobiotechnology spans materials science, biology, medicine, environmental science, and tech, these disparate research and regulatory disciplines must come together, often for the first time—to fully understand the impact on humans and the environment.
Dual use: Good for us, bad for us
Like other quickly growing fields, there’s a time lag between the promise of new advances and the possibilities of unintended uses.
“There were so many more applications than we thought there were,” said Bradley, who collected exciting nanobio examples such as Alzheimer’s treatment, permanent contact lenses, organ replacement, and enhanced muscle recovery, among others.
The article also highlights concerns about crossing the blood-brain barrier, thought-initiated control of computers, and nano-enabled DNA editing where the researchers suggest more caution, questioning, and attention could be warranted. This attention spans everything from deep fundamental research and regulations all the way to what Omberg called “the equivalent of tattoo removal” if home-DNA splicing attempts go south.
The researchers draw parallels to more established fields such as synthetic bio and pharmacology, which offer lessons to be learned from current concerns such as the unintended consequences of fentanyl and opioids. They believe these fields also offer examples of innovative coordination between science and ethics, such as synthetic bio’s IGEM [The International Genetically Engineered Machine competition]—student competition, to think about not just how to create, but also to shape the use and control of new technologies.
Omberg said unusually enthusiastic early reviewers of the article contributed even more potential uses and concerns, demonstrating that experts in many fields recognize ethical nanobiotechnology is an issue to get in front of. “This is a train that’s going. It will be sad if 10 years from now, we haven’t figured how to talk about it.”
Funding for the team’s research was supported by PNNL’s Biorisk Beyond the List National Security Directorate Objective.
Here’s a link to and a citation for the paper,
The Promise of Emergent Nanobiotechnologies for In Vivo Applications and Implications for Safety and Security by Anne M. Arnold, Ashley M. Bradley, Karen L. Taylor, Zachary C. Kennedy, and Kristin M. Omberg. Health Security.Oct 2022.408-423.Published in Volume: 20 Issue 5: October 17, 2022 DOI: https://doi.org/10.1089/hs.2022.0014 Published Online:17 Oct 2022
This paper is open access.
You can find out more about IGEM (The International Genetically Engineered Machine competition) here.
Comments (brief)
It seems a little odd that the news release (“Prosthetics moved by thoughts …”) and the paper both reference neurotechnology without ever mentioning it by name. Here’s the reference from the paper, Note: Links have been removed,
Nanoparticles May Be Developed to Facilitate Cognitive Enhancements
The development and implementation of NPs that enhance cognitive function has yet to be realized. However, recent advances on the micro- and macro-level with neural–machine interfacing provide the building blocks necessary to develop this technology on the nanoscale. A noninvasive brain–computer interface to control a robotic arm was developed by teams at 2 universities.157 A US-based company, Neuralink, [emphasis mine] is at the forefront of implementing implantable, intracortical microelectrodes that provide an interface between the human brain and technology.158,159 Utilization of intracortical microelectrodes may ultimately provide thought-initiated access and control of computers and mobile devices, and possibly expand cognitive function by accessing underutilized areas of the brain.158
Neuralink (founded by Elon Musk) is controversial for its animal testing practices. You can find out more in Björn Ólafsson’s May 30, 2023 article for Sentient Media.
The focus on nanoparticles as the key factor in the various technologies and applications mentioned seems narrow but necessary given the breadth of topics covered in the paper as the authors themselves note in the paper’s abstract,
… In this article, while not comprehensive, we attempt to illustrate the breadth and promise of bionanotechnology developments, and how they may present future safety and security challenges. Specifically, we address current advancements to streamline the development of engineered NPs for in vivo applications and provide discussion on nano–bio interactions, NP in vivo delivery, nanoenhancement of human performance, nanomedicine, and the impacts of NPs on human health and the environment.
…
They have a good overview of the history and discussions about nanotechnology risks and regulation. It’s international in scope with a heavy emphasis on US efforts, as one would expect.
For anyone who’s interested in the neurotechnology end of things, I’ve got a July 17, 2023 commentary “Unveiling the Neurotechnology Landscape: Scientific Advancements, Innovations and Major Trends—a UNESCO report.” The report was launched July 13, 2023 during UNESCO’s Global dialogue on the ethics of neurotechnology (see my July 7, 2023 posting about the then upcoming dialogue for links to more UNESCO information). Both the July 17 and July 7, 2023 postings included additional information about Neuralink.