With all the focus on COVID-19, viruses , and aerosols, it’s easy to forget that there are other kinds of contaminated air too. The last time I featured work on nanoparticles and air pollution was in a May 31, 2017 posting, “Explaining the link between air pollution and heart disease?” where scientists announced they may have discovered how air pollution (nanoparticles) were making their way from lungs to the heart. Answer: the bloodstream.
A July 3, 2020 Lancaster University press release (also on EurekAlert) announces research into how air made toxic by metallic nanoparticles affects the heart in very specific ways (Note: A link has been removed),
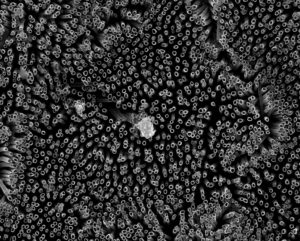
Toxic metallic air pollution nanoparticles are getting inside the crucial, energy-producing structures within the hearts of people living in polluted cities, causing cardiac stress – a new study confirms.
The research team, led by Professors Barbara Maher of Lancaster University and Lilian Calderón-Garcidueñas of The University of Montana and the Universidad del Valle de Mexico, found the metallic nanoparticles, which included iron-rich nanoparticles and other pollution-derived metals such as titanium, inside the damaged heart cells of a 26-year-old and even a three-year-old toddler.
The hearts had belonged to people who had died in accidents and who had lived in highly-polluted Mexico City.
The findings shed new light on how air pollution can cause the development of heart disease, as the iron-rich particles were associated with damage to the cells, and increased cardiac oxidative stress, even in these very young hearts.
The repeated inhalation of these iron-rich nanoparticles, and their circulation by the bloodstream to the heart, may account for the well-established associations between exposure to particulate air pollution and increased cardiovascular disease, including heart attacks. The study indicates that heart disease can start in very early age, before progressing to full-blown cardiovascular illness later in life. This type of air pollution may thus be responsible for the ‘silent epidemic’ of heart disease, internationally. By causing pre-existing heart conditions, it may also account for some of the increased death rates from Covid-19 seen in areas with high levels of particulate air pollution.
Professor Maher said: “It’s been known for a long time that people with high exposure to particulate air pollution experience increased levels and severity of heart disease. Our new work shows that iron-rich nanoparticles from air pollution can get right inside the millions of mitochondria inside our hearts…the structures which generate the energy needed for our hearts to pump properly.
“That we found these metal particles inside the heart of even a three-year old indicates that we’re setting heart disease in train right from the earliest days, but only seeing its full, clinical effects in later life. It’s really urgent to reduce emissions of ultrafine particles from our vehicles and from industry, before we give heart disease to the next generation too.”
The researchers, using high-resolution transmission electron microscopy and energy-dispersive X-ray analysis, found that the mitochondria containing the iron-rich nanoparticles appeared to be damaged, with some cells showing deformities and others with ruptured membranes. Professor Calderón-Garcidueñas stated that increased levels of markers of cardiac oxidative stress are present in the very young cases examined.
The iron-rich nanoparticles found inside the heart cells are identical in size, shape and composition to those emitted from sources such as the exhausts, tyres and brakes of vehicles. These air pollution nanoparticles are also emitted by industrial sources as well as open fires in homes.
Some of the iron-rich nanoparticles are also strongly magnetic. This raises concerns about what might happen when people with millions of these nanoparticles in their hearts are using appliances with associated magnetic fields, such as hair dryers and mobile phones. People who work in industries that mean they are exposed to magnetic fields such as welders and power line engineers may also be at risk. This kind of exposure could potentially lead to heart electrical dysfunction and cell damage.
The findings builds on the researchers’ previous findings that show that the hearts of city dwellers contain billions of these nanoparticles and can be up to ten times more polluted than the hearts of people living in less polluted places.
The researchers say their study underlines the need for governments across the world to tackle ultrafine particulate pollution in their cities.
Professor Calderón-Garcidueñas said: “Exposure to such air pollution is a modifiable risk factor for cardiovascular disease, on a global scale, reinforcing the urgent need for individual and government actions not just to reduce PM2.5 but to monitor, regulate and reduce emissions of these specific, ultrafine components of the urban air pollution ‘cocktail’.”
Here’s a link to and a citation for the paper,
Iron-rich air pollution nanoparticles: An unrecognised environmental risk factor for myocardial mitochondrial dysfunction and cardiac oxidative stress by B.A.Maher, A.González-Maciel, R.Reynoso-Robles, R.Torres-Jardón, L.Calderón-Garcidueñas. Environmental Research Volume 188, September 2020, 109816 DOI: https://doi.org/10.1016/j.envres.2020.109816 Available online 21 June 2020
This paper appears to be open access (just keep scrolling down).